ATP functions as a pathogen-associated molecular pattern to activate the E3 ubiquitin ligase RNF213
Juraj Ahel, Arda Balci, Victoria Faas, Daniel B. Grabarczyk, Roosa Harmo, Daniel R. Squair, Jiazhen Zhang, Elisabeth Roitinger, Frederic Lamoliatte, Sunil Mathur, Luiza Deszcz, Lillie E. Bell, Anita Lehner, Thomas L. Williams, Hanna Sowar, Anton Meinhart, Nicola T. Wood, Tim Clausen, Satpal Virdee, Adam J. Fletcher
Nature Communications
Highlights
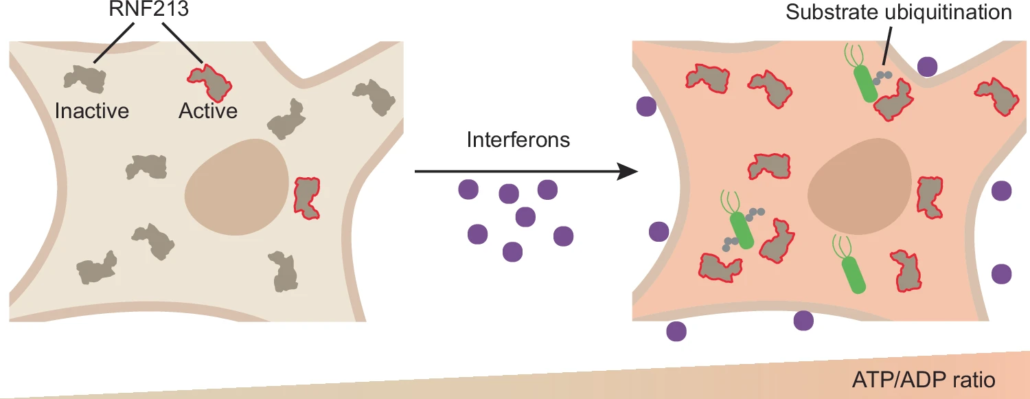
- RNF213 is an E3 ubiquitin ligase activated by ATP binding, crucial for broad antimicrobial immunity.
- Cellular ATP levels, increased by interferons, reversibly regulate RNF213 E3 ligase activity in living cells.
- RNF213 uses a unique ATP-dependent transthiolation mechanism involving a key catalytic cysteine in its RZ domain.
- ATP acts as an intracellular danger signal activating RNF213, representing a new class of ATP-regulated E3 enzymes.
Read the original paper here