ZNFX1 uses two-component ubiquitin circuitry to quarantine viral RNA
Daniel R. Squair1,‡, Eilidh Rivers2,‡, Hanna Sowar2, Arda Balci2, Roosa Harmo2, David J. Wright1, Gaurav Beniwal1, Mathieu Soetens1, Sunil Mathur1, Aidan Tollervey2, Nicola T. Wood1, Kuan-Chuan Pao1, Callum Stanton1, Adam J. Fletcher2,* and Satpal Virdee1,*
Molecular Cell
Highlights
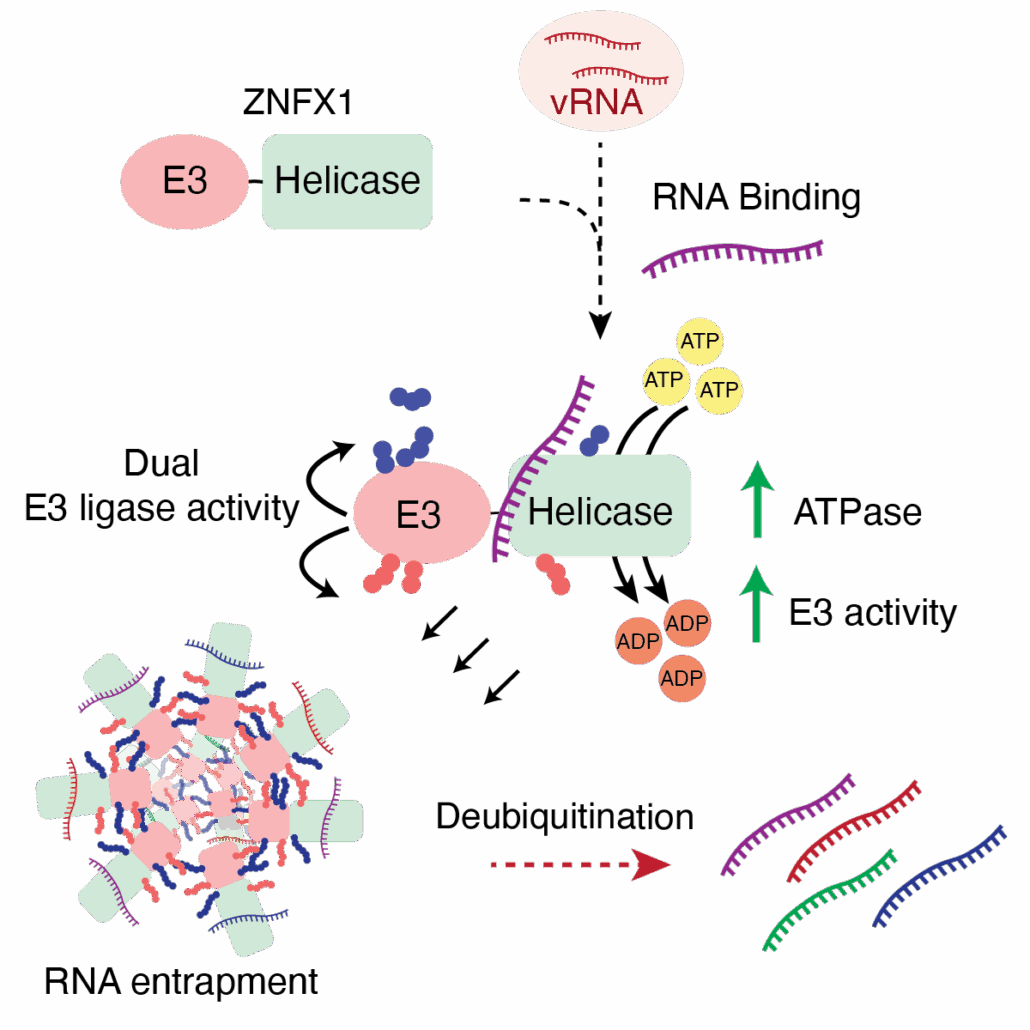
- Activity-based E3 ligase profiling identifies ZNFX1 as an atypical E3 ligase
- ATP-dependent RNA binding to the SF1 helicase domain activates dual E3 ligase function
- E3 activity and ATP-dependent RNA binding are required for broad viral restriction
- ZNFX1 drives ubiquitin-regulated RNA sequestration into self-propagating aggregates
Read the original paper here